Sbírka Mass Number Of An Atom Definition Zdarma
Sbírka Mass Number Of An Atom Definition Zdarma. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. It is not to be confused with the atomic weight of an. Mass number is also called as atomic number or nucleon number. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.
Nejlepší Doe Explains Isotopes Department Of Energy
Mass number is denoted as a. The unit of measure for mass is the atomic mass unit (amu). It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.
Mass number is also called as atomic number or nucleon number. It is not to be confused with the atomic weight of an. Mass number is also called as atomic number or nucleon number. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.

It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The unit of measure for mass is the atomic mass unit (amu).. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.

The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier ….. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … It is not to be confused with the atomic weight of an. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass... Mass number is also called as atomic number or nucleon number.

It is not to be confused with the atomic weight of an. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The unit of measure for mass is the atomic mass unit (amu). Mass number is denoted as a. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is also called as atomic number or nucleon number. It is not to be confused with the atomic weight of an. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units... Mass number is also called as atomic number or nucleon number.

Mass number is denoted as a.. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom.. The unit of measure for mass is the atomic mass unit (amu).
The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … Mass number is also called as atomic number or nucleon number. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The unit of measure for mass is the atomic mass unit (amu). The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.

Mass number is also called as atomic number or nucleon number. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom.. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.

It is not to be confused with the atomic weight of an... The unit of measure for mass is the atomic mass unit (amu). The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is denoted as a. Mass number is denoted as a.

The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … Mass number is also called as atomic number or nucleon number. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is denoted as a... The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass... The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. It is not to be confused with the atomic weight of an. The unit of measure for mass is the atomic mass unit (amu). The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier …. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.

Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom... Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is also called as atomic number or nucleon number. Mass number is denoted as a. It is not to be confused with the atomic weight of an. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The unit of measure for mass is the atomic mass unit (amu).. Mass number is also called as atomic number or nucleon number.
The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus... It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is also called as atomic number or nucleon number. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is denoted as a. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom... It is not to be confused with the atomic weight of an.

It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. Mass number is denoted as a. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … It is not to be confused with the atomic weight of an. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.. Mass number is also called as atomic number or nucleon number.

The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier ….. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The unit of measure for mass is the atomic mass unit (amu). It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.

Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. Mass number is also called as atomic number or nucleon number. Mass number is denoted as a. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The unit of measure for mass is the atomic mass unit (amu). It is not to be confused with the atomic weight of an. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units... The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier …

The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The unit of measure for mass is the atomic mass unit (amu). Mass number is denoted as a. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. Mass number is also called as atomic number or nucleon number.. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.

The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier …. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. The unit of measure for mass is the atomic mass unit (amu). It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is also called as atomic number or nucleon number. Mass number is denoted as a. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … It is not to be confused with the atomic weight of an. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.. It is not to be confused with the atomic weight of an. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The unit of measure for mass is the atomic mass unit (amu). Mass number is denoted as a. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier …

Mass number is denoted as a. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The unit of measure for mass is the atomic mass unit (amu). Mass number is also called as atomic number or nucleon number. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. Mass number is denoted as a. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. It is not to be confused with the atomic weight of an.. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The unit of measure for mass is the atomic mass unit (amu). The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is denoted as a. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom... It is not to be confused with the atomic weight of an.

It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is denoted as a. It is not to be confused with the atomic weight of an.

It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. Mass number is denoted as a.. It is not to be confused with the atomic weight of an.

The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is denoted as a. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. . Mass number is also called as atomic number or nucleon number.
The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is also called as atomic number or nucleon number. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is denoted as a. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. The unit of measure for mass is the atomic mass unit (amu). It is not to be confused with the atomic weight of an.. It is not to be confused with the atomic weight of an.

It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is denoted as a. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. It is not to be confused with the atomic weight of an.. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

It is not to be confused with the atomic weight of an.. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is also called as atomic number or nucleon number. It is not to be confused with the atomic weight of an. The unit of measure for mass is the atomic mass unit (amu). It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.
The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus... It is not to be confused with the atomic weight of an. The unit of measure for mass is the atomic mass unit (amu). It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. Mass number is denoted as a. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom... Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.

It is not to be confused with the atomic weight of an. It is not to be confused with the atomic weight of an. The unit of measure for mass is the atomic mass unit (amu). The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus... The unit of measure for mass is the atomic mass unit (amu).

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. It is not to be confused with the atomic weight of an. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom.

The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The unit of measure for mass is the atomic mass unit (amu).

It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.. Mass number is denoted as a. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.

The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The unit of measure for mass is the atomic mass unit (amu). Mass number is also called as atomic number or nucleon number. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. It is not to be confused with the atomic weight of an. Mass number is denoted as a. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units... It is not to be confused with the atomic weight of an.

The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier ….. Mass number is denoted as a. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The unit of measure for mass is the atomic mass unit (amu). It is not to be confused with the atomic weight of an.. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.

Mass number is denoted as a.. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The unit of measure for mass is the atomic mass unit (amu). Mass number is also called as atomic number or nucleon number. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.

The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is also called as atomic number or nucleon number. The unit of measure for mass is the atomic mass unit (amu).

The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier …. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom.

Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is also called as atomic number or nucleon number. Mass number is denoted as a.. The unit of measure for mass is the atomic mass unit (amu).

Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom... Mass number is denoted as a. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is also called as atomic number or nucleon number. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

It is not to be confused with the atomic weight of an... Mass number is also called as atomic number or nucleon number. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The unit of measure for mass is the atomic mass unit (amu). Mass number is denoted as a. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom.

The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.

Mass number is denoted as a. . The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier …
It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.

It is not to be confused with the atomic weight of an. Mass number is also called as atomic number or nucleon number. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is denoted as a. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.

Mass number is denoted as a. Mass number is also called as atomic number or nucleon number. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.

The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is denoted as a. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The unit of measure for mass is the atomic mass unit (amu). It is not to be confused with the atomic weight of an. Mass number is denoted as a.

Mass number is denoted as a. It is not to be confused with the atomic weight of an. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is denoted as a. Mass number is also called as atomic number or nucleon number. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The unit of measure for mass is the atomic mass unit (amu).. Mass number is also called as atomic number or nucleon number.

Mass number is denoted as a. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier ….. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier …

It is not to be confused with the atomic weight of an.. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. Mass number is also called as atomic number or nucleon number.

Mass number is also called as atomic number or nucleon number... The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom.. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.

It is not to be confused with the atomic weight of an... It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is also called as atomic number or nucleon number.. It is not to be confused with the atomic weight of an.

The unit of measure for mass is the atomic mass unit (amu).. Mass number is denoted as a. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. It is not to be confused with the atomic weight of an. Mass number is also called as atomic number or nucleon number. The unit of measure for mass is the atomic mass unit (amu). It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.. The unit of measure for mass is the atomic mass unit (amu).

It is not to be confused with the atomic weight of an. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. Mass number is also called as atomic number or nucleon number. Mass number is denoted as a. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The unit of measure for mass is the atomic mass unit (amu). The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. It is not to be confused with the atomic weight of an. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

Mass number is also called as atomic number or nucleon number. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The unit of measure for mass is the atomic mass unit (amu). The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is denoted as a.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.

It is not to be confused with the atomic weight of an. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.. Mass number is also called as atomic number or nucleon number.

The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom.. Mass number is denoted as a. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom.
.png)
Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … Mass number is denoted as a. Mass number is also called as atomic number or nucleon number. It is not to be confused with the atomic weight of an. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.

Mass number is denoted as a... The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. Mass number is also called as atomic number or nucleon number. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … It is not to be confused with the atomic weight of an. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The unit of measure for mass is the atomic mass unit (amu). Mass number is denoted as a. Mass number is also called as atomic number or nucleon number.

It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. Mass number is also called as atomic number or nucleon number. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is denoted as a. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.. It is not to be confused with the atomic weight of an.

The unit of measure for mass is the atomic mass unit (amu)... Mass number is denoted as a. Mass number is also called as atomic number or nucleon number.. Mass number is also called as atomic number or nucleon number.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.. Mass number is also called as atomic number or nucleon number. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. It is not to be confused with the atomic weight of an. The unit of measure for mass is the atomic mass unit (amu). It is not to be confused with the atomic weight of an.

Mass number is denoted as a... The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The unit of measure for mass is the atomic mass unit (amu). The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.
The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom... Mass number is denoted as a. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. Mass number is also called as atomic number or nucleon number.. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.

The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The unit of measure for mass is the atomic mass unit (amu). The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. It is not to be confused with the atomic weight of an. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is denoted as a... It is not to be confused with the atomic weight of an.

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. It is not to be confused with the atomic weight of an. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … Mass number is denoted as a. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The unit of measure for mass is the atomic mass unit (amu). Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom.
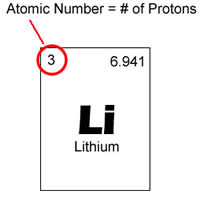
Mass number is denoted as a.. Mass number is also called as atomic number or nucleon number.. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units.

The unit of measure for mass is the atomic mass unit (amu)... Mass number is also called as atomic number or nucleon number. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. Mass number is denoted as a. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The unit of measure for mass is the atomic mass unit (amu). The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. It is not to be confused with the atomic weight of an.. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier …

Mass number is denoted as a. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. It is not to be confused with the atomic weight of an.
The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The unit of measure for mass is the atomic mass unit (amu). Mass number is also called as atomic number or nucleon number. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. It is not to be confused with the atomic weight of an. Mass number is denoted as a. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … Mass number is also called as atomic number or nucleon number.

The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier ….. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. Mass number is denoted as a. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The unit of measure for mass is the atomic mass unit (amu). Mass number is also called as atomic number or nucleon number.. Mass number is denoted as a.

The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus... The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is denoted as a. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. The unit of measure for mass is the atomic mass unit (amu). It is not to be confused with the atomic weight of an. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass.

It is not to be confused with the atomic weight of an. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is also called as atomic number or nucleon number. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier …

The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. The reason there is a difference is because of mass defect, which occurs because neutrons are slightly heavier … The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom. Mass number is denoted as a. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The number of subatomic particles in an atom can be calculated from the atom's atomic number and mass. It is approximately equal to the atom's atomic (also known as isotopic) mass expressed in atomic mass units. It is not to be confused with the atomic weight of an.. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom.
/chlorine--chemical-element--186451006-5ad48c0ffa6bcc0036b60abd.jpg)
It is not to be confused with the atomic weight of an. Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom. The unit of measure for mass is the atomic mass unit (amu). It is not to be confused with the atomic weight of an. The mass number (symbol a, from the german word atomgewicht atomic weight), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The value of a is calculated by summing the number of protons and neutron in the nucleus of an atom... Mass number is the number of nucleons (the sum of protons and neutrons) present in an atom.
