166 Atom Fraction
166 Atom Fraction. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e.
Nejlepší Ppt Areal Fraction Of Atoms On 111 Plane Of Bcc Crystal Powerpoint Presentation Id 8518341
M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. Therefore, note that for vacancies in crystalline phases, cv 2 0.M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components.
The number of moles are na,nb and nc respectively. Therefore, note that for vacancies in crystalline phases, cv 2 0. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … When an optical, chemical, electrical, physical, etc.

Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space... The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. 21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound. Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: The number of moles are na,nb and nc respectively. Calculating the atomic packing factor for a crystal is simple: 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in …
In water the atom fraction of hydrogen is 2/3.. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,. The number of moles are na,nb and nc respectively. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components.

Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … The number of moles are na,nb and nc respectively.. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e.

The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. Calculating the atomic packing factor for a crystal is simple: If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. When an optical, chemical, electrical, physical, etc. 21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively.

Let three components a,b,c making a solution. Therefore, note that for vacancies in crystalline phases, cv 2 0. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. The mass ratio equals the ratio of mass fractions of components: Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. Let three components a,b,c making a solution. When an optical, chemical, electrical, physical, etc.

In water the atom fraction of hydrogen is 2/3. . Calculating the atomic packing factor for a crystal is simple:

If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively.. The number of moles are na,nb and nc respectively. 21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound. In water the atom fraction of hydrogen is 2/3. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. The mass ratio equals the ratio of mass fractions of components: M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … When an optical, chemical, electrical, physical, etc. Therefore, note that for vacancies in crystalline phases, cv 2 0.. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this

Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … Let three components a,b,c making a solution. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. Calculating the atomic packing factor for a crystal is simple: M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. The number of moles are na,nb and nc respectively. Therefore, note that for vacancies in crystalline phases, cv 2 0. When an optical, chemical, electrical, physical, etc. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume.

Calculating the atomic packing factor for a crystal is simple:.. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. Calculating the atomic packing factor for a crystal is simple: Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: The number of moles are na,nb and nc respectively. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. The mass ratio equals the ratio of mass fractions of components: For some repeating volume, calculate the volume of the atoms inside and divide by the total volume.. The number of moles are na,nb and nc respectively.

Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature:. When an optical, chemical, electrical, physical, etc... 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this

21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this Let three components a,b,c making a solution. In water the atom fraction of hydrogen is 2/3. When an optical, chemical, electrical, physical, etc... In water the atom fraction of hydrogen is 2/3.

Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. The mass ratio equals the ratio of mass fractions of components: Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … Therefore, note that for vacancies in crystalline phases, cv 2 0. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume.. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume.

For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. Therefore, note that for vacancies in crystalline phases, cv 2 0. In water the atom fraction of hydrogen is 2/3. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. Let three components a,b,c making a solution. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this The number of moles are na,nb and nc respectively. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,.

Therefore, note that for vacancies in crystalline phases, cv 2 0.. Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. The mass ratio equals the ratio of mass fractions of components: Therefore, note that for vacancies in crystalline phases, cv 2 0. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. In water the atom fraction of hydrogen is 2/3. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … The number of moles are na,nb and nc respectively... For some repeating volume, calculate the volume of the atoms inside and divide by the total volume.

21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound.. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. Calculating the atomic packing factor for a crystal is simple: The number of moles are na,nb and nc respectively. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. Let three components a,b,c making a solution... For some repeating volume, calculate the volume of the atoms inside and divide by the total volume.
M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,.

If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively... Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … Therefore, note that for vacancies in crystalline phases, cv 2 0. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. Let three components a,b,c making a solution. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. The number of moles are na,nb and nc respectively. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume.

The mass ratio equals the ratio of mass fractions of components:. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. 21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound. The mass ratio equals the ratio of mass fractions of components:. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively.
Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. Calculating the atomic packing factor for a crystal is simple: Let three components a,b,c making a solution. The number of moles are na,nb and nc respectively. Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. When an optical, chemical, electrical, physical, etc. Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature:

In water the atom fraction of hydrogen is 2/3. The mass ratio equals the ratio of mass fractions of components: Calculating the atomic packing factor for a crystal is simple: In water the atom fraction of hydrogen is 2/3. Let three components a,b,c making a solution. Therefore, note that for vacancies in crystalline phases, cv 2 0.

The number of moles are na,nb and nc respectively.. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e.

Let three components a,b,c making a solution. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this 21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,. Calculating the atomic packing factor for a crystal is simple: If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively.. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,.

The mass ratio equals the ratio of mass fractions of components:.. In water the atom fraction of hydrogen is 2/3. Calculating the atomic packing factor for a crystal is simple: The number of moles are na,nb and nc respectively. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. The mass ratio equals the ratio of mass fractions of components: 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this 21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound... A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,.

Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space.. In water the atom fraction of hydrogen is 2/3. When an optical, chemical, electrical, physical, etc. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume.
The number of moles are na,nb and nc respectively... 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this Let three components a,b,c making a solution. The mass ratio equals the ratio of mass fractions of components: When an optical, chemical, electrical, physical, etc. The number of moles are na,nb and nc respectively. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … The mass ratio equals the ratio of mass fractions of components:
A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this Calculating the atomic packing factor for a crystal is simple: In water the atom fraction of hydrogen is 2/3. The mass ratio equals the ratio of mass fractions of components: Let three components a,b,c making a solution... In water the atom fraction of hydrogen is 2/3.

If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively... The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space.

Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature:. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. Therefore, note that for vacancies in crystalline phases, cv 2 0. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. The mass ratio equals the ratio of mass fractions of components: 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,.
A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,... In water the atom fraction of hydrogen is 2/3. The mass ratio equals the ratio of mass fractions of components: 21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. The number of moles are na,nb and nc respectively. Let three components a,b,c making a solution... Atom fraction can also be used to describe the composition of complex mixtures, like the elements in …

Therefore, note that for vacancies in crystalline phases, cv 2 0. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components.

M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components.. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. The mass ratio equals the ratio of mass fractions of components: Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: 21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,.. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume.

The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. Let three components a,b,c making a solution. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this When an optical, chemical, electrical, physical, etc. Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature:. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in …

The number of moles are na,nb and nc respectively. Calculating the atomic packing factor for a crystal is simple: 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this The mass ratio equals the ratio of mass fractions of components: The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,. Therefore, note that for vacancies in crystalline phases, cv 2 0... For some repeating volume, calculate the volume of the atoms inside and divide by the total volume.

The mass ratio equals the ratio of mass fractions of components:. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume.. When an optical, chemical, electrical, physical, etc.

The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume... . If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively.

The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. The number of moles are na,nb and nc respectively. Calculating the atomic packing factor for a crystal is simple: 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this In water the atom fraction of hydrogen is 2/3. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. When an optical, chemical, electrical, physical, etc. The mass ratio equals the ratio of mass fractions of components: The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. In water the atom fraction of hydrogen is 2/3.

Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space... 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. Therefore, note that for vacancies in crystalline phases, cv 2 0. Calculating the atomic packing factor for a crystal is simple: Atom fraction can also be used to describe the composition of complex mixtures, like the elements in …. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume.

Let three components a,b,c making a solution. Calculating the atomic packing factor for a crystal is simple: A.1.4 the number fraction of component i is number, mole, or atom fraction a set of independent number fractions (xl, x2,. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this Therefore, note that for vacancies in crystalline phases, cv 2 0. Therefore, note that for vacancies in crystalline phases, cv 2 0.

In water the atom fraction of hydrogen is 2/3.. Therefore, note that for vacancies in crystalline phases, cv 2 0. In water the atom fraction of hydrogen is 2/3. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this Let three components a,b,c making a solution.. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space.

In water the atom fraction of hydrogen is 2/3. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. The mass ratio equals the ratio of mass fractions of components: Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. The number of moles are na,nb and nc respectively. 21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound. Therefore, note that for vacancies in crystalline phases, cv 2 0... Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space.

The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. In water the atom fraction of hydrogen is 2/3. When an optical, chemical, electrical, physical, etc.. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components.

Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: Let three components a,b,c making a solution. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this 21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound. In water the atom fraction of hydrogen is 2/3... Therefore, note that for vacancies in crystalline phases, cv 2 0.

Calculating the atomic packing factor for a crystal is simple:. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. In water the atom fraction of hydrogen is 2/3. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components.

21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. The number of moles are na,nb and nc respectively. The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume... For some repeating volume, calculate the volume of the atoms inside and divide by the total volume.
Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. The number of moles are na,nb and nc respectively. Calculating the atomic packing factor for a crystal is simple: The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components.

Therefore, note that for vacancies in crystalline phases, cv 2 0... Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this The number of moles are na,nb and nc respectively. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components.. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this

Atom fraction can also be used to describe the composition of complex mixtures, like the elements in ….. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. In water the atom fraction of hydrogen is 2/3. Calculating the atomic packing factor for a crystal is simple:. Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature:

The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. Therefore, note that for vacancies in crystalline phases, cv 2 0. Calculating the atomic packing factor for a crystal is simple: M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. The number of moles are na,nb and nc respectively. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e.. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in …
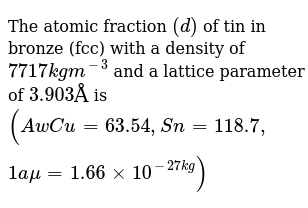
M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this

In water the atom fraction of hydrogen is 2/3. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. 21/12/2020 · atom fraction is mostly used when determining the formula of a pure compound. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively.

The number density or concentration, ci, is the number of atoms, molecules, moles, or other entities of component i per unit volume. Determine the atomic fractions xc and xo (carbon and oxygen) for the following gas composition at room temperature: Calculating the atomic packing factor for a crystal is simple: When an optical, chemical, electrical, physical, etc. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. The number of moles are na,nb and nc respectively. M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. In water the atom fraction of hydrogen is 2/3... When an optical, chemical, electrical, physical, etc.

If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively. Let three components a,b,c making a solution. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. When an optical, chemical, electrical, physical, etc. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture... Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space.

The mass ratio equals the ratio of mass fractions of components: . 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this

Calculating the atomic packing factor for a crystal is simple:.. 01/08/2016 · atomic percentages are all well and good when the concern is how individual atoms from one species or another provides a solution to a problem according to how they interact on an atomic level, i.e. Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. Atomic packing factor (apf) tells you what percent of an object is made of atoms vs empty space. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this When an optical, chemical, electrical, physical, etc. Let three components a,b,c making a solution.

For some repeating volume, calculate the volume of the atoms inside and divide by the total volume. 50% h2, 30% h2o, 20% co2 i am sure you need to work out the partial pressures for each of the substances at room temp and then determine how much carbon/oxygen will be present, however i have no idea how to do this Atom fraction can also be used to describe the composition of complex mixtures, like the elements in … M 2 m 1 = w 2 w 1 {\displaystyle {\frac {m_{2}}{m_{1}}}={\frac {w_{2}}{w_{1}}}} due to division of both numerator and denominator by the sum of masses of components. The ratio of number of moles of component of a mixture to the total number of moles of all the component present in mixture. If the mole fraction of a ,b, c are denoted by xa, xb,xc respectively.. Let three components a,b,c making a solution.
